Our collaboration on ancestry-specific biases in Tumor Mutational Burden (TMB) now out (Nassar, Adib, Alaiwi et al. Cancer Cell) 🎉
Our study of the reliability of tumor-only estimates of Tumor Mutational Burden (TMB) and it’s influence on immunotherapy outcomes across ancestries is now out in Cancer Cell. This work was led by Amin Nassar, Elio Adib, and Sarah Abou Alaiwi, and in collaboration with Jian Carrot-Zhang at MSKCC:
Ancestry-driven recalibration of tumor mutational burden and disparate clinical outcomes in response to immune checkpoint inhibitors.
Nassar A[+], Adib E[+], Alaiwi S[+], Zarif T, Groha S, Akl E, Nuzzo P, Mouhieddine T, Perea-Chamblee T, Taraszka K, El-Khoury H, Labban M, Fong C, Arora KS, Labaki C, Xu W, Sonpavde G, Haddad R, Mouw K, Giannakis M, Hodi F, Zaitlen N, Schoenfeld AJ, Schultz N, Berger MF, MacConaill L, Ananda G, Kwiatkowski D, Choueiri T, Schrag D, Carrot-Zhang J[+], Gusev A[+]. Cancer Cell. 2022
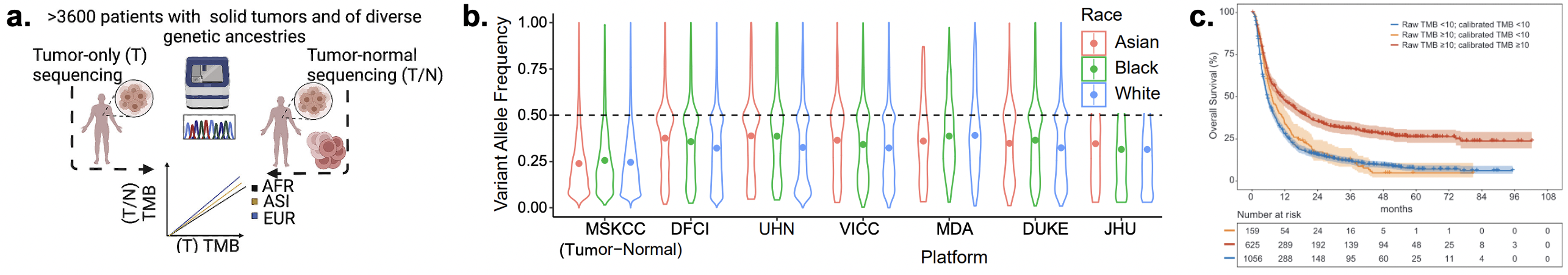
Having a tumor with high TMB is an FDA approved indicator for immunotherapy, but are current approaches to estimate TMB reliable and generalizable? In this study, we showed TMB estimates from tumor-only sequencing can be biased, with a substantially greater bias in individuals having non-European genetic ancestry. Importantly, this increased bias is not due to any differences in tumor biology, but a direct consequence of tumor-only filtering using smaller reference panels for non-Europeans. We propose a correction for this bias based on estimates of global ancestry, and we show that falsely categorized high TMB patients do not respond better to immunotherapy. Pooling together data from two institutions, and having applied our correction, we explore the ancestry-specific effect of TMB on immunotherapy outcomes as well as implications for individual driver genes. Our study highlights how disparities in one dataset (here, germline reference panels) can lead to unexpected disparities in seemingly unrelated data, such as somatic variant calling.
For more, see the twitter thread from co-first author Amin Nassar, the accompanying highlight (Snyder and McGranahan, Cancer Cell), as well as press in Science, Nature, The New Scientist, and EurekAlert.
Ancestry-specific TMB biases | (a) Study design. (b) Systematic biases in Variant Allele Frequency by race for tumor-only sequencing (inflation in tumors from non-white patients). (c) No survival benefit for patients with recalibrated TMB calls.